Flazalone
 | |
| Clinical data | |
|---|---|
| Other names | Flumefenine, R-760. |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
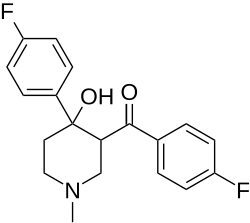
| Formula | C19H19F2NO2 |
| Molar mass | 331.363 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Flazalone is an anti-inflammatory drug that has not been approved as a medicine.[1][2][3]
According to Shaomeng Wang and co-workers, replacement of the para-fluoro halogen with a meta,para-dichloro substitution resulted in dopamine reuptake inhibitors useful in treating cocaine addiction.[4][5]
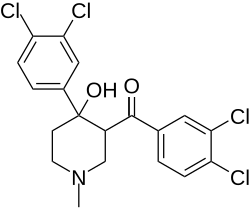
Diclazalone (Flazalone analog)
Synthesis
The synthesis has been covered:[6][7] Patents:[8][9] Alternate synthesis:[10] 62%:[11]
N.B. The p-fluoroacetophenone [403-42-9] has dual use in the synthesis of Enecadin [259525-01-4].
See also
References
- ↑ Levy L, McClure D (August 1976). "The pharmacology of flazalone: a new class of anti-inflammatory agent". The Journal of Pharmacology and Experimental Therapeutics. 198 (2): 473–480. doi:10.1016/S0022-3565(25)30615-4. PMID 7668.
- ↑ "Flazalone". Inxight Drugs. The National Center for Advancing Translational Sciences (NCATS).
- ↑ Castañer, J., Arrigoni-Martelli, E. (1977). "Flazalone". Drugs of the Future. 2 (3): 169. doi:10.1358/dof.1977.002.03.998237. ISSN 0377-8282.
- ↑ Wang S, Sakamuri S, Enyedy IJ, Kozikowski AP, Deschaux O, Bandyopadhyay BC, et al. (February 2000). "Discovery of a novel dopamine transporter inhibitor, 4-hydroxy-1-methyl-4-(4-methylphenyl)-3-piperidyl 4-methylphenyl ketone, as a potential cocaine antagonist through 3D-database pharmacophore searching. Molecular modeling, structure-activity relationships, and behavioral pharmacological studies". Journal of Medicinal Chemistry. 43 (3): 351–60. doi:10.1021/jm990516x. PMID 10669562.
- ↑ Wang S, Sakamuri S, Enyedy IJ, Kozikowski AP, Zaman WA, Johnson KM (July 2001). "Molecular modeling, structure--activity relationships and functional antagonism studies of 4-hydroxy-1-methyl-4-(4-methylphenyl)-3-piperidyl 4-methylphenyl ketones as a novel class of dopamine transporter inhibitors". Bioorganic & Medicinal Chemistry. 9 (7): 1753–64. doi:10.1016/s0968-0896(01)00090-6. PMID 11425577.
- ↑ Lednicer, D., Mitscher, L. A. (1980). The organic chemistry of drug synthesis. 2. Wiley. ISBN 9780471043928.
- ↑ Draper, M. D., Petracek, F. J., Klohs, M. W., McClure, D. A., Levy, L., Ré, O. N. (October 1972). "Fluorophenyl-4-(p-fluorophenyl)-4-hydroxy-1-methyl-3-piperidyl ketone (flazalone): a novel non-steroidal anti-inflammatory agent". Arzneimittel-Forschung. 22 (10): 1803. PMID 4677080.
- ↑ Daniel Draper Marshall, CH496700 & NL6811811 (1970 to Rexall Drug Chemical).
- ↑ M Draper & L Levy, U.S. patent 3,849,578 (1974 to Riker Laboratories Inc).
- ↑ Charles M Leir, U.S. patent 3,887,568 (1975 to Riker Laboratories Inc).
- ↑ Stenlake, J. (December 1989). "Synthèse et étude préliminaire de quelques arylpropanonamines substitués sur le cycle et leurs sels quaternaires". European Journal of Medicinal Chemistry. 24 (6): 591–597. doi:10.1016/0223-5234(89)90026-3. ISSN 0223-5234.